Dendritic cells (DCs), so called because of their penetrating, “tree-like” or dendritic forms, are in charge of the start of adaptive immune responses and serve as the immune system’s “guards.” In 1868, Paul Langerhans initially identified DCs in human skin, however, he mistakenly identified them as cutaneous nerve cells. Leukocytes generated from bone marrow (BM) called DCs are the most effective antigen-presenting cells. Utilising possible varieties of growth factors, like granulocyte macrophage-colony stimulating factor (GM-CSF), they can also be generated in vitro from BM and blood.
Human dendritic cells have been developed to collect and break down antigens into peptides that can be displayed on MHC molecules that T cells can recognise. However all DC subtypes are responsible for antigen uptake, handling, and appearance to naive T cells, DCs are heterogeneous, including myeloid and plasmacytoid DCs. The DC subtypes vary in location, migration patterns pathways, specific immunological function, and reliance on infections or inflammatory stimuli for their production. The phenotype and activity of DCs are critical for establishing tolerance and memory throughout the maturation of an adaptive immune response.
How Do Dendritic Cells Connect Adaptive & Innate Immunity?
Due to their numerous cytoplasmic processes, DCs have a vast surface area that enables close contact with a variety of nearby cells, including neutrophils, epithelial cells, T cells, and natural killer cells. For instance, in experiments, 100–3000 T cells can be stimulated by a single mature DC. Nearly every non-lymphoid tissue receives a migration of DC precursors from the BM through the bloodstream, where they remain in an immature stage and continuously sample their surroundings through endocytosis, macropinocytosis, and phagocytosis.
Even in the absence of obvious infection or inflammation, they can expand their pathways through the tight junctions of epithelia to improve antigen uptake. When a disease invades a tissue, local iDCs use pattern recognition receptors (like TLRs) to identify intruders, capture antigens and immediately exit the area. In reaction to various chemokines like CCL19 and CCL21, they scurry across the cells, cross the endothelium of lymphatic arteries, and go to the draining lymph nodes (LN).
DCs go through phenotypical and functional development as they move away from the peripheral tissues. The most amazing thing about them is that they stop collecting antigens while simultaneously up-regulating the activity of co-stimulatory markers like CD80 and CD86 and the chemokine receptor CCR7, and secreting pro-inflammatory cytokines like TNF- and IL-12. DCs travel to T-cell zones after entering the subcapsular sinus of the lymph node. The transmission of antigens to T cells in this instance is actively facilitated by the dendritic cells.
Role in Immunotherapy
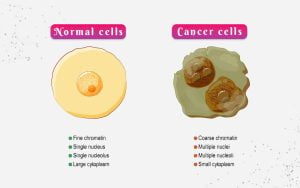
Using dendritic cells’ immune-regulatory abilities to treat cancer, and autoimmune conditions, and avoid transplant rejection offers enormous promise. Human dendritic cells may be modified to become the most powerful adjuvant to boost the host’s immunological defences. Tumours have been found to inhibit DCs in cases of cancer by secreting anti-inflammatory cytokines like IL-10, which then primes the local DCs to develop suppressive T lymphocytes. DCs might be created outside of the body, loaded with tumour antigens, and then injected back into the body to increase the host’s defences against the cancerous cells.
These DC vaccines are often safe, have few adverse effects, and have shown to be practical and efficient in some individuals. Other methods of using DCs to treat a variety of illnesses have been described and are currently being tested in clinical trials.