The production of blood as long as an organism’s life is orchestrated by hematopoietic stem cells (HSCs). Accordingly, HSCs form each type of blood cell that has varying functions such as red blood cells that carry oxygen to the immune cells that protect our body from pathogen attack to platelets that mediate clotting. There are various genes involved in the self-renewal of HSCs and differentiation into various cell types. The aorto-gonado-mesonephros region of the embryo is the first site where HSCs can be recognized after which the cells reside in the bone marrow in mammals.
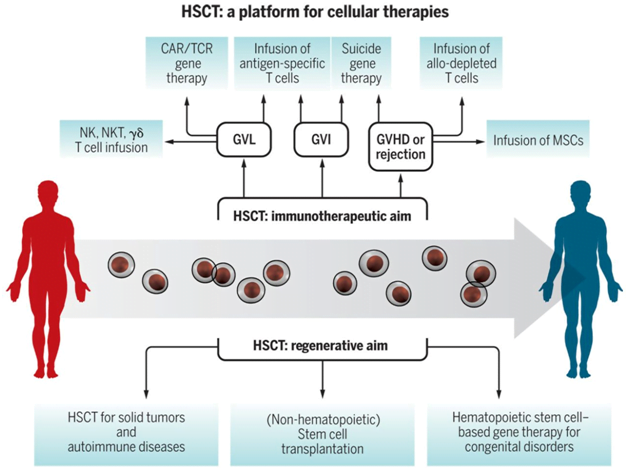
The transplantation of bone marrow involves the transplantation of adult stem cells (HLA-matched) or cord blood cells into a recipient. The process of hematopoietic stem cell transplantation (HSCT) was the first approach of regenerative therapy to make its entry for treatment. It is a cell transplantation approach used on a large scale given the hundreds of clinics using the approach (Christian Chabannon et al, 2018).
In the case of allo-HSCT or using allogeneic cells for transplantation, the use of cells from the bone marrow, peripheral blood and cord blood can allow transplants from both related and unrelated donors. The use of reduced-intensity conditioning that limits the use of radiation can allow even the young to access the therapies. Additionally, post-transplant the administration of cyclophosphamide as an immunosuppressant can allow for overcoming the early belief that 100% HLA match is required for allogeneic HSCT.
The use of autologous HSCT coupled with immunosuppression improved clinical conditions such as severe multiple sclerosis and inflammatory diseases as shown by many global multicenter studies (Christian Chabannon et al, 2018).
The development of HSCs administered with genetically engineered T lymphocytes allowed for the treatment of genetic diseases such as adenosine deaminase severe combined immunodeficiency (ADA-SCID), hemoglobinopathies and neurodegenerative diseases. For example, Glaxo Smith Kline (GSK) developed Strimvelis® made of genetically modified autologous hematopoietic stem cells for ADA-SCID that received approval by the EU. This is the first HSC-based gene therapy product to receive approval from the European Medicines Agency (EMA) making the field of HSCs here to stay!
The use of the latest technologies such as gene editing offers promise in cell therapies. For example, gene editing involves the introduction of breaks in double-stranded DNA to edit the genome. The use of transcription activator-like effector nuclease (TALEN)–mediated gene editing and the introduction of CD19 (CAR19) or the B-cell antigen on T-cells in 2 infants with relapsed refractory CD19+ B cell acute lymphoblastic leukemia showed remission in 28 days. (Qasim et al, Science Translational Medicine, 2017).
HSCT has completed 60 years and is still a largely used Stem Cell Therapy. The use of cutting edge technology can further augment the use of HSCs for treating dreadful diseases that strike people of all ages across the globe. HSCs, as they say, “old is gold” are here to stay and weave their magic.
References:
Ng A, Alexander W. Haematopoietic stem cells: past, present and future. Cell Death Discov. 3, 17002 (2017).
- Qasim, H. Zhan, S. Samarasinghe et al, Molecular remission of infant B-ALL after infusion of universal TALEN gene-edited CAR T cells. Science Translational Medicine. 9, eaaj2013 (2017).
Christian Chabannon, Jurgen Kuball, Attilio Bondanza, Et Al. Hematopoietic Stem Cell Transplantation In Its 60s: A Platform For Cellular Therapies. Science Translational Medicine 11 Apr 2018