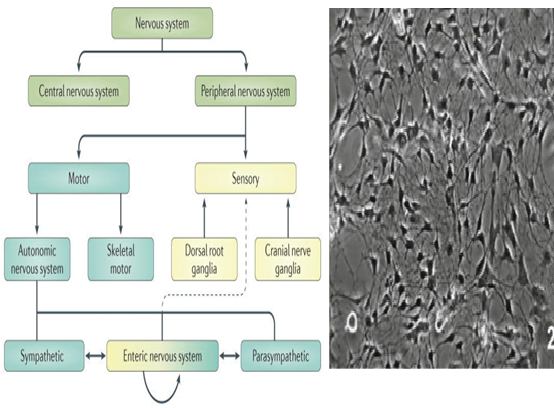
The gut performs a wide array of functions ranging from digestion and absorption across different compartments along the gut. Additionally, there is a precise activity of skeletal muscles and smooth muscles that allow the contractions of the gut. The secretion of mucus, enzymes and salts occurs at correct timing and intervals. The epithelial barrier between the lumen and the inner environment not only absorbs but also does not allow the leakage of materials in the intestine such as the enzymes or bacteria into the inner environment. There are also immune cells in the barrier that maintain the immunity of the gut. The Gastrointestinal Nerves or Enteric Nervous System (ENS) regulates all these activities. This system is composed of 100 million neurons and usually functions in sync with the central nervous system (CNS).
However, research published by Rao and Gershon in 2016 in Nature reviews Gastroenterology & hepatology, states that the presence of intrinsic microcircuits in the ENS can allow this system to work autonomously without the CNS. Also, the anatomy, signaling circuits, and neurotransmitters are the same in both the CNS and ENS with many CNS diseases affecting the gut!
For example, research in The Journal of Autism and Developmental Disorders by Marler reviewed that the blood of autism spectrum disorder (ASD) patients had higher levels of serotonin (5-HT) that is secreted by the gut. This was connected to the gastrointestinal nerves symptoms such as stool retention or stomach pain seen in these patients. The report also showed the latency of varicella-zoster virus (VZV; the causative agent of chickenpox and shingles) in many neurons including the neurons of the Gastrointestinal Nerves. A mutation called Gly93Ala in superoxide dismutase (SOD1) in mice associated with Amyotrophic lateral sclerosis (a neurodegenerative disease) shows damaged Paneth cells of the gut!
Things get even more interesting when Hsiao and team showed that the host gut microbiota is vital in the “leaky gut” seen in autism spectrum disorder (ASD). The 2013-published Cell article showed that the intestine was repaired when Bacteroides fragilis was administered to an ASD-mice model that also improved several symptoms of ASD in the mice. This opens up the possibility of addressing ASD by targeting the ENS.
These multiple implications of the ENS have earned it the name “the second brain”. In order to develop a system to study the ENS further, researchers, Zhang and Hu published an article in Methods in molecular biology to establish a primary culture of mouse ENS neurons. Few points for successful culture suggested are given below:
- Collagenase was first used to separate the smooth muscle layer and myenteric plexus; the latter was treated with trypsin. Culture conditions allow the survival of only the neurons and glia with the smooth muscle cells dying.
- The collagenase digestion step should be checked every 5 minutes and should be terminated when the tissue becomes white.
- As the myenteric plexus is between muscles, contamination from the lumen is lowered by removing the mucosa and sub-mucosa.
- Attachment of neurons is facilitated by the use of poly-D-lysine/laminin coating or Matrigel™ matrix coating on coverslips that allow the successful culture.
Thus, such primary cultures of the “second brain” can allow the study of physiology and chemical molecules used to treat diseases. Ultimately, the research aims at addressing the root cause of diseases that can be facilitated by such cultures.
References:
Rao, M., & Gershon, M. D. (2016). The bowel and beyond: the enteric nervous system in neurological disorders. Nature reviews. Gastroenterology & hepatology, 13(9), 517–528.
Marler S, et al. Brief report: whole blood serotonin levels and gastrointestinal nerves symptoms in autism spectrum disorder. The Journal of Autism and Developmental Disorders. 2016;46:1124–1130.
Hsiao EY, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. 2013;155:1451–1463.
Zhang, Y., & Hu, W. (2013). Mouse enteric neuronal cell culture. Methods in molecular biology (Clifton, N.J.), 1078, 55–63.